Search results for: “Search”
-
107 Nobel Laureate Attack on Greenpeace Traced Back to Biotech PR Operators
by Jonathan Latham, PhD (Independent Science News) Greenpeace was denied entrance yesterday (June 30) to a National Press Club Event in Washington, DC of 107 Nobel Laureates. The event was ostensibly organised by a scientific group calling itself Support Precision Agriculture to publicise a letter signed by 107 Nobel Laureates demanding that Greenpeace cease its […]
-

This scientist is trying to stop a lab-created global disaster
Disclaimer: This article is interesting as it expresses the concerns of a scientist deeply involved in the synbio and in particular gene-drive fields (his team figured out how to make dene-drives work!), and highlights just how under-regulated current experiments are. We don’t condone the research described in the article, or necessarily agree with the alternative […]
-
Meet the New Stevia! GMOs 2.0 Get Dressed for Success
by Stacy Malkan (The Huffington Post) Our culture is smitten with the notion that technology can save us – or at least create great business opportunities! Cargill, for example, is working on a new food technology that mimics stevia, a sugar substitute derived from plant leaves, for the “exploding sports nutrition market.” Cargill’s new product, […]
-

The National Academies’ Gene Drive study has ignored important and obvious issues
by Jim Thomas (The Guardian): ‘Gene drives’ seem to be the ultimate high-leverage technology. Yesterday’s report from the US National Academies begun the job of describing what is at stake, but it missed some important questions. If there is a prize for the fastest emerging tech controversy of the century the ‘gene drive’ may have […]
-
Permanently changing a species: What could go wrong?
by Dana Perls, Food and Technology campaigner (Friends of the Earth US) No commercial or environmental release of gene drives, says Friends of the Earth The National Academies of Sciences released a new report today, which calls for robust safety assessments for “gene drive modified organisms.” The NAS says the controversial new genetic engineering technology […]
-
Stop The Gene Bomb! ETC Group Comment on NAS Report on Gene Drives
The National Academy of Sciences is releasing a report about “gene drives”, which are attempts to cause entire populations to inherit certain traits, such as reducing reproductive capacity, and which can cause species crashes, or even extinction. ETC Group have countered with this news release on the topic: 8th June 2016 · First study on […]
-
Mascoma: The biggest misspending of public funds for cellulosic biofuels ever?
This is an investigation of Mascoma Corporation, a start-up biofuels company which may have misspent more public funds intended for building advanced biofuels refineries than any other company in North America. Download the investigation Summary: Mascoma took at least $100m and possibly over $155m in public funding intended for building integrated biorefineries. Their biggest donor […]
-

Latin American scientists warn against risks of new GM techniques
GM Watch: Scientists demand an end to experimentation in genetic manipulation and call on farmers and society to reclaim scientific research Below is an important statement by the Latin American Union of Scientists Concerned with Society and Nature, questioning the safety of new GM techniques like gene editing and demanding the suspension of experimentation in […]
-
U.S. tries to export its biotech deregulatory regime
by Dr. Steve Suppan (Institute for Agriculture and Trade Policy) A long standing claim by the U.S. government and agribusiness lobby is that U.S. regulations on genetically engineered (GE) crops are science-based while European regulations are not. For example, an April 8 letter from the American Soybean Association to the U.S. Trade Representative Michael Froman […]
-
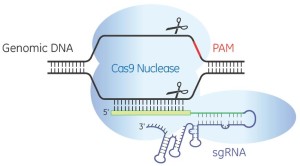
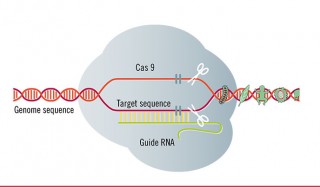
God’s Red Pencil? CRISPR and The Three Myths of Precise Genome Editing
by Jonathan Latham, PhD (Independent Science News) For the benefit of those parts of the world where public acceptance of biotechnology is incomplete, a public relations blitz is at full tilt. It concerns an emerging set of methods for altering the DNA of living organisms. “Easy DNA Editing Will Remake the World. Buckle Up“; “We […]